Содержание
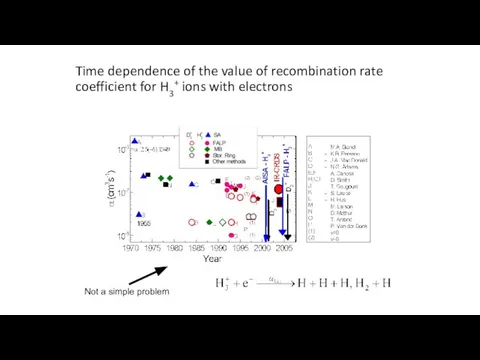
- 2. Time dependence of the value of recombination rate coefficient for H3+ ions with electrons Not a
- 3. Different experiments Ion Storage Ring Afterglow plasma + No buffer gas + Excellent energy resolution Complicated
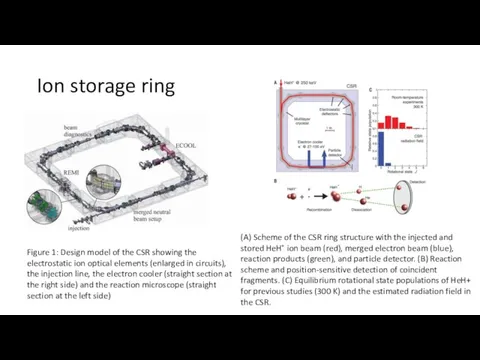
- 4. Ion storage ring Figure 1: Design model of the CSR showing the electrostatic ion optical elements
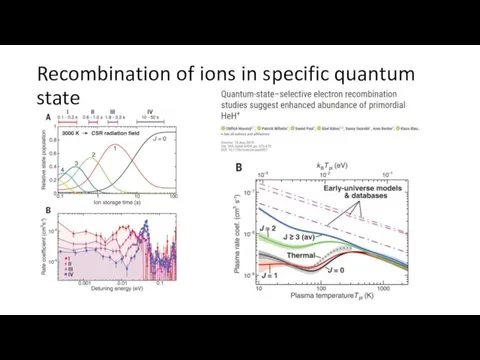
- 5. Recombination of ions in specific quantum state
- 6. Rekombination of H3+ ions with electrons Neutral assisted ternary recombination: Electron assisted ternary recombination: Formation of
- 7. Stationary afterglow plasma Combination of Stationary afterglow and absorption spectroscopy
- 8. Stationary afterglow plasma AISA – Advanced Integrated Stationary Afterglow Mass spectrometer + Langmuir probe diagnostics Phys.
- 9. Stationary afterglow plasma Microwave diagnostics + mass spectrometry
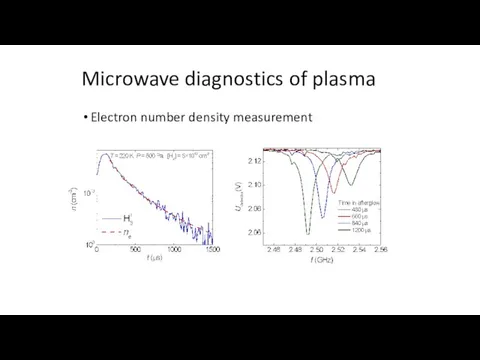
- 10. Microwave diagnostics of plasma Plasma conductivity Change of resonant frequency and resonator quality: Plasma frequency
- 11. Microwave diagnostics of plasma Sicha et al., Czech. J. Phys. B 20, 684, 1970 From the
- 12. Electron number density measurement Microwave diagnostics of plasma
- 13. SA-CRDS apparatus CRDS – Cavity Ring Down Spectroscopy SA – Stationary Afterglow Highly reflective mirror Discharge
- 14. Cavity ringdown spectroscopy First used for mirror reflectivity determination (Herbelin et al. 1980). Later, the dependence
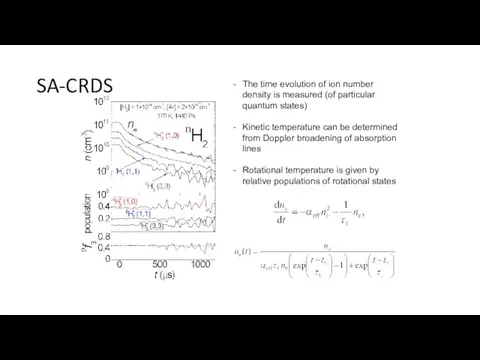
- 15. SA-CRDS The time evolution of ion number density is measured (of particular quantum states) Kinetic temperature
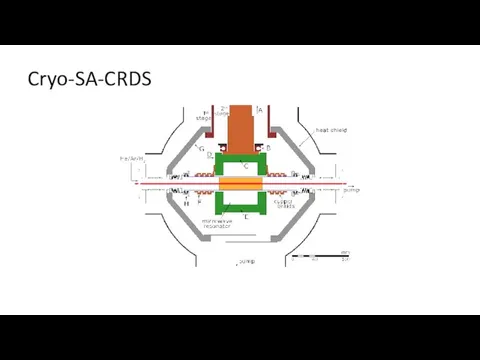
- 16. Cryo-SA-CRDS
- 17. Cryo-SA-CRDS
- 18. Cryo-SA-CRDS Plašil, R; Dohnal, P; Kálosi, Á; Roučka, Š; Shapko, D; Rednyk, S; Johnsen, R; Glosík,
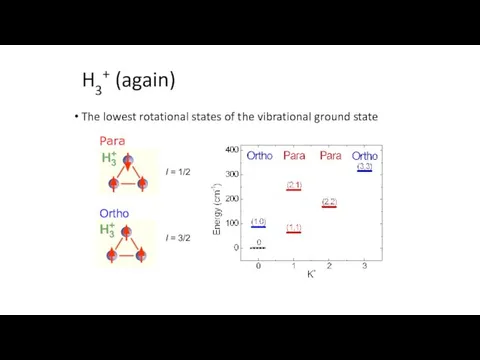
- 19. H3+ (again) The lowest rotational states of the vibrational ground state Para I = 1/2 I
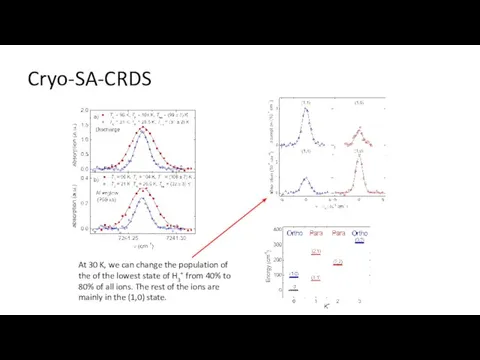
- 20. Cryo-SA-CRDS At 30 K, we can change the population of the of the lowest state of
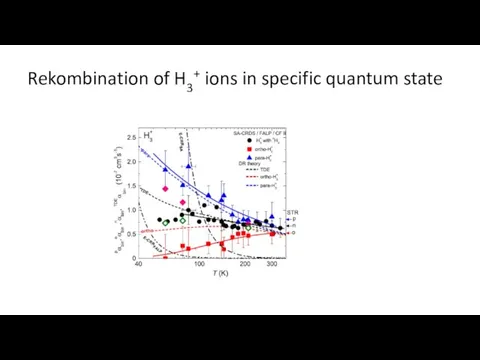
- 21. Rekombination of H3+ ions in specific quantum state
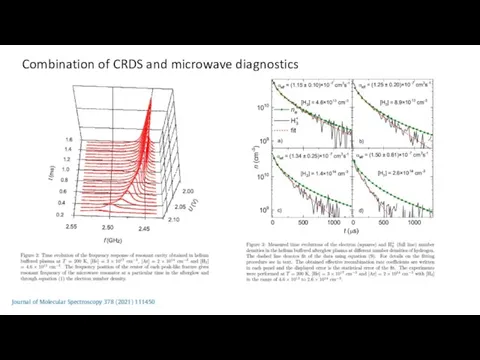
- 22. Combination of CRDS and microwave diagnostics
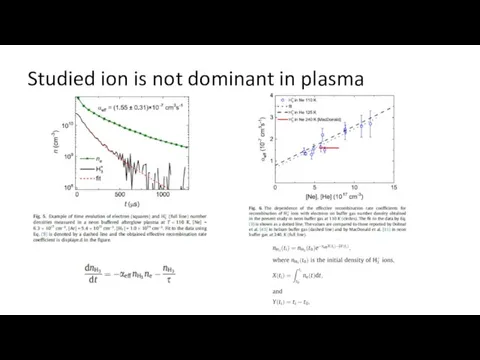
- 23. Studied ion is not dominant in plasma
- 24. Flowing afterglow plasma
- 25. Flowing afterglow plasma
- 26. FALP, SIFT, SIFDT FALP – Flowing Afterglow with Langmuir Probe SIFT – Selective Ion Flow Tube
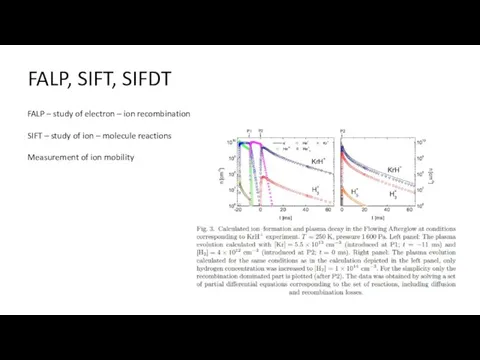
- 27. FALP, SIFT, SIFDT FALP – study of electron – ion recombination SIFT – study of ion
- 28. Cryo-FALP II Cryogenic Flowing Afterglow with Langmuir Probe Flowtube diameter – 5 cm He buffer gas
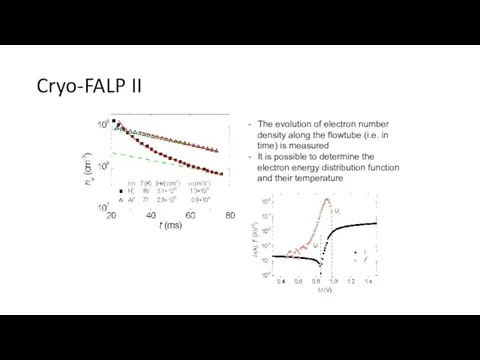
- 29. Cryo-FALP II The evolution of electron number density along the flowtube (i.e. in time) is measured
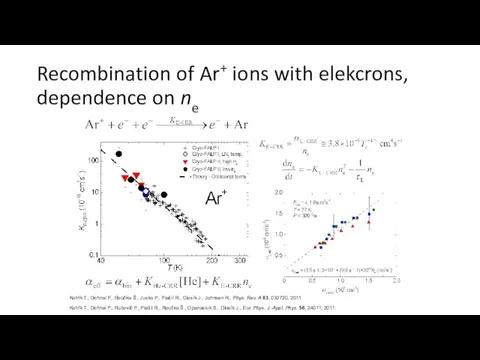
- 30. Recombination of Ar+ ions with elekcrons, dependence on ne Kotrík T., Dohnal P., Roučka Š., Jusko
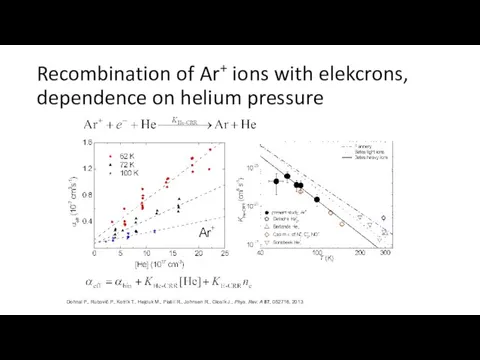
- 31. Recombination of Ar+ ions with elekcrons, dependence on helium pressure Dohnal P., Rubovič P., Kotrík T.,
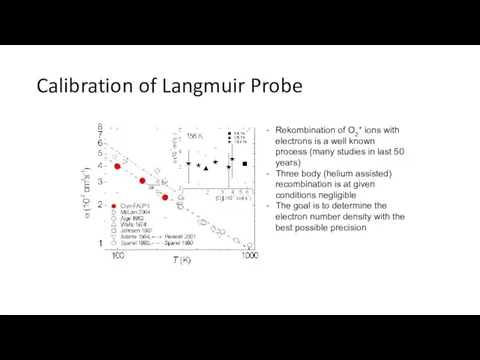
- 32. Calibration of Langmuir Probe Rekombination of O2+ ions with electrons is a well known process (many
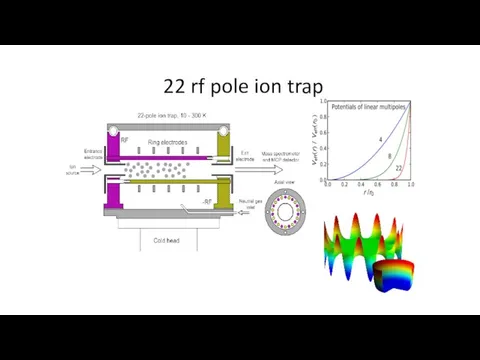
- 33. 22 rf pole ion trap Many configurations for different experiments Cold Heads at 22PT and H
- 34. 22 rf pole ion trap
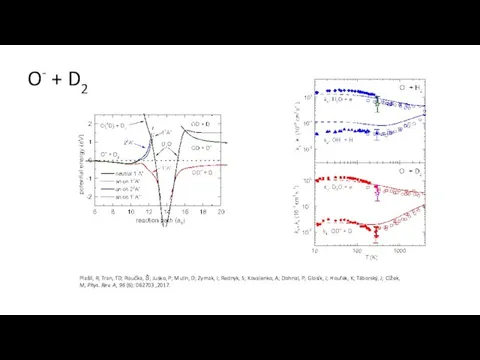
- 35. O- + D2 Plašil, R; Tran, TD; Roučka, Š; Jusko, P; Mulin, D; Zymak, I; Rednyk,
- 37. Скачать презентацию
















 Тезаурус по атомной энергетике
Тезаурус по атомной энергетике Дефект массы
Дефект массы Электрические свойства веществ - классификация
Электрические свойства веществ - классификация Тепловые двигатели
Тепловые двигатели Зубчатый механизм
Зубчатый механизм Демонтаж системы питания инжекторных и карбюраторных двигателей
Демонтаж системы питания инжекторных и карбюраторных двигателей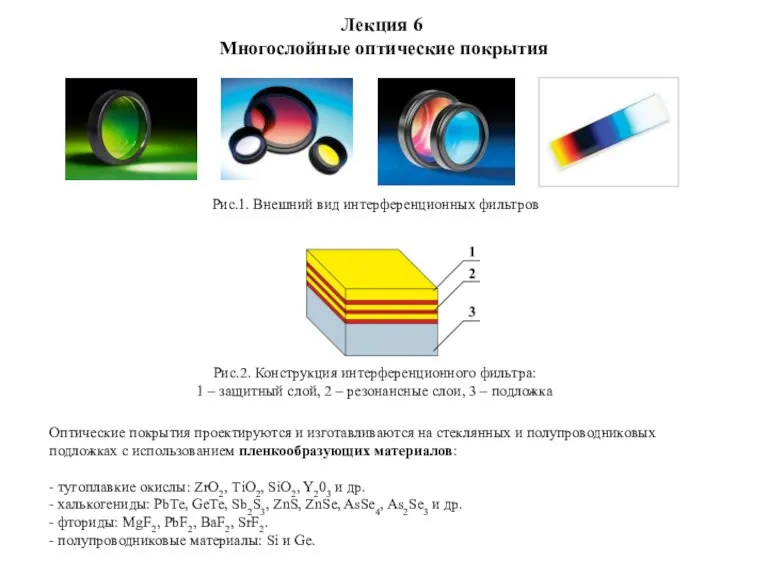 Многослойные оптические покрытия
Многослойные оптические покрытия Радиационный контроль
Радиационный контроль Презентация на тему Конденсаторы и электроёмкость
Презентация на тему Конденсаторы и электроёмкость  Волны. Типы волн
Волны. Типы волн Спектральный анализ
Спектральный анализ Как сберечь тепло в доме
Как сберечь тепло в доме Магнитное поле. Электромагнитная индукция. Подготовка к контрольной работе
Магнитное поле. Электромагнитная индукция. Подготовка к контрольной работе Измерение физических величин
Измерение физических величин Лазер на свободных электронах. Разеры, гразеры
Лазер на свободных электронах. Разеры, гразеры Абрикосов Алексей Алексеевич
Абрикосов Алексей Алексеевич Презентация на тему Солнце и другие звезды
Презентация на тему Солнце и другие звезды  Плотность веществ. Задачи
Плотность веществ. Задачи Роль света в жизни человека
Роль света в жизни человека Учение М.В. Ломоносова о свете и цвете в контексте натурфилософии и художественных исканий середины XVII века
Учение М.В. Ломоносова о свете и цвете в контексте натурфилософии и художественных исканий середины XVII века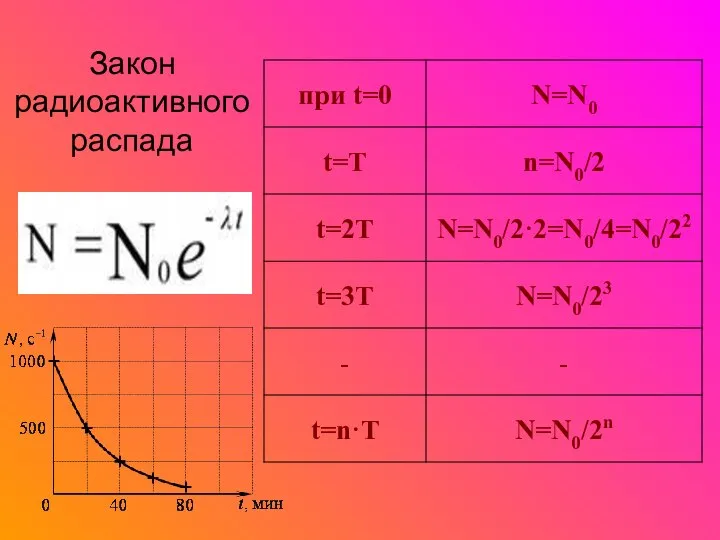 Закон радиоактивного распада
Закон радиоактивного распада Газовые законы
Газовые законы Кавитация. Кавитация в ЦБН: последствия
Кавитация. Кавитация в ЦБН: последствия Свойства твёрдых тел, жидкостей и газов
Свойства твёрдых тел, жидкостей и газов Закон всесвітнього тяжіння. Сила тяжіння
Закон всесвітнього тяжіння. Сила тяжіння Презентация на тему Колебания
Презентация на тему Колебания  Эволюция развития реакторов
Эволюция развития реакторов Потери напряжения в элементах электрической цепи
Потери напряжения в элементах электрической цепи