Содержание
- 2. http://www.earthday.gov/ Today is Earth Day, April 22nd 2003
- 3. It isn’t the pollution that’s harming the environment. It’s the impurities in our air and water
- 4. ENERGY STAR - Energy-efficient choices can save families about a third on their energy bill with
- 5. Learn to Conserve Water in Your Home - You can also take a virtual tour that
- 6. Ecosystems an assemblage of different species and their physical environment, all organized in a way that
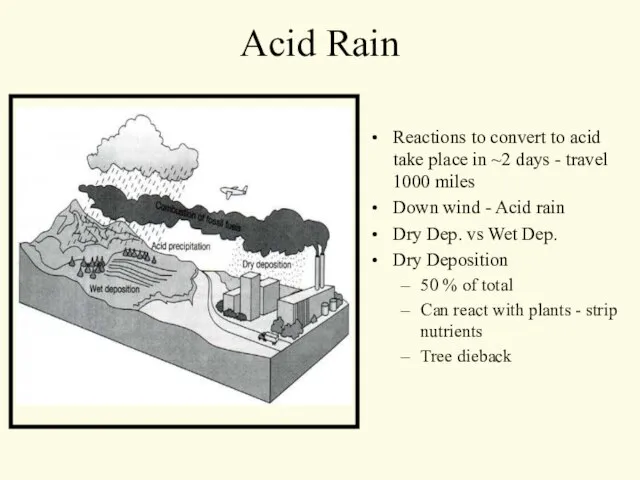
- 7. Acid Rain Reactions to convert to acid take place in ~2 days - travel 1000 miles
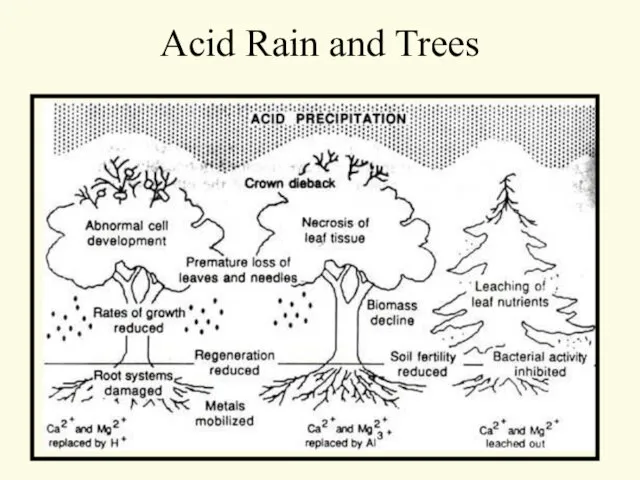
- 9. Acid Rain and Trees
- 10. Forests affected by Acid Rain Northeast US Canada Northern Europe Asia
- 11. Acid Rain and Buildings Many buildings are made of concrete and or stone These compounds act
- 12. Europe The US Capitol
- 13. Human Emissions - Combustion NOx N2 +O2 --> 2NO Forms in high temperatures of combustion engine
- 14. Human Emissions - Fertilizer N2 +Energy H+--> NH3 Formed by the Haber process Added to fields
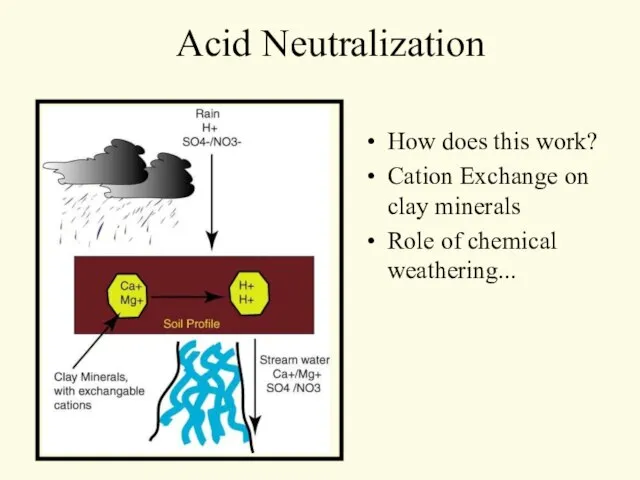
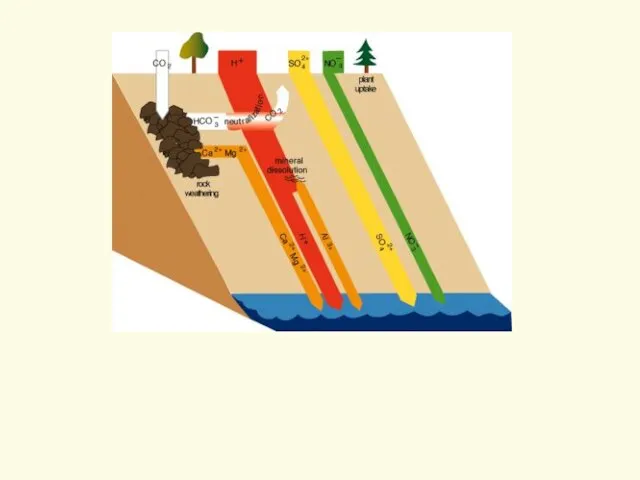
- 15. Acid Neutralization How does this work? Cation Exchange on clay minerals Role of chemical weathering...
- 16. Where do N emissions originate? ~ 55% come from agriculture ~ 25% come from industry –
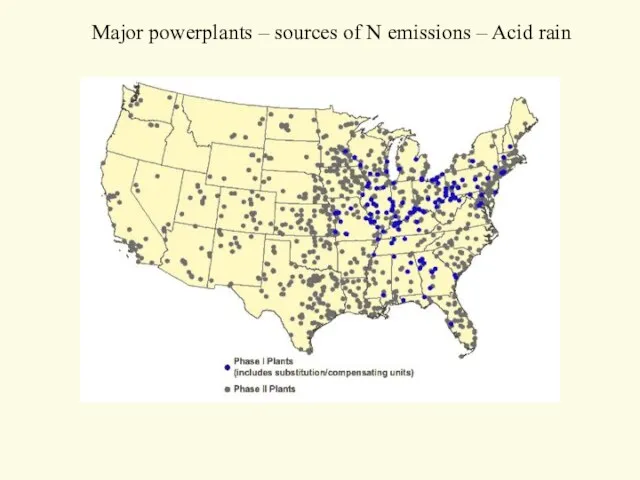
- 17. Major powerplants – sources of N emissions – Acid rain
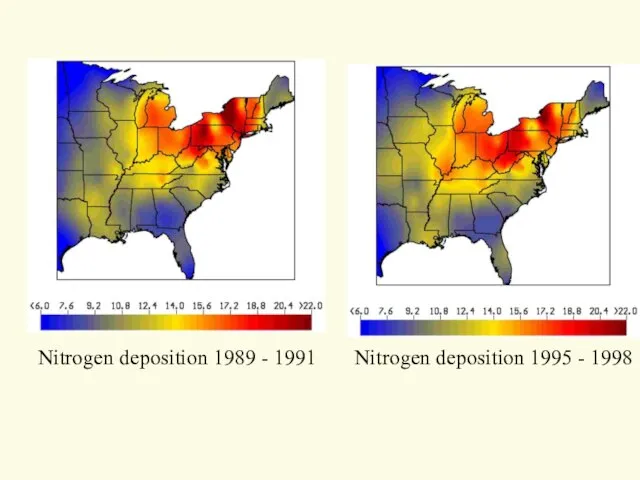
- 18. Nitrogen deposition 1989 - 1991 Nitrogen deposition 1995 - 1998
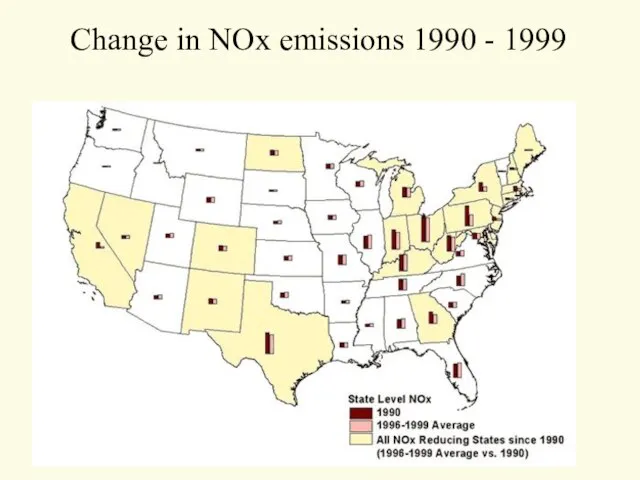
- 19. Change in NOx emissions 1990 - 1999
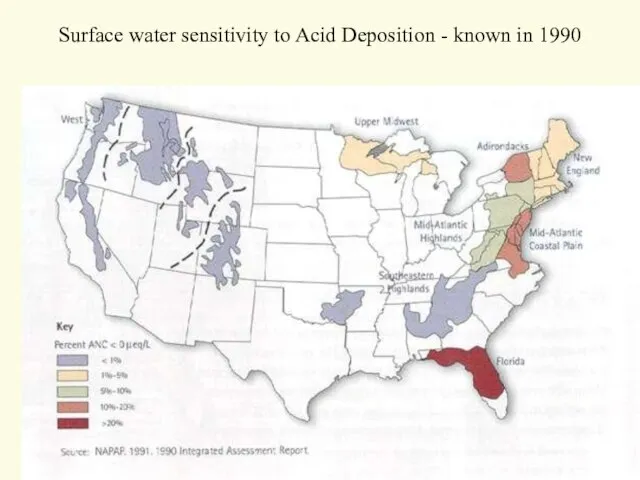
- 20. Surface water sensitivity to Acid Deposition - known in 1990
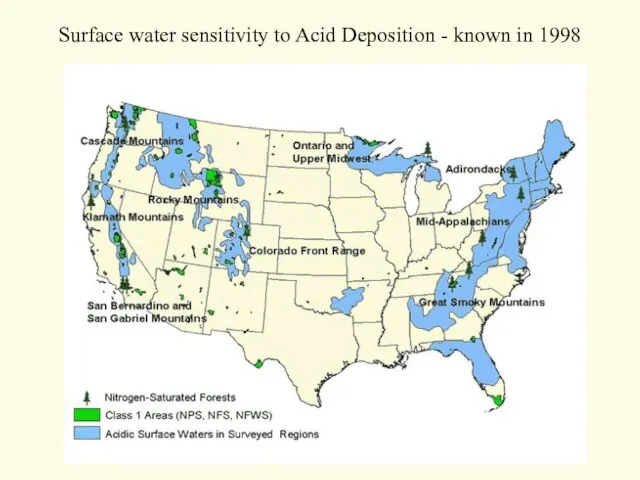
- 21. Surface water sensitivity to Acid Deposition - known in 1998
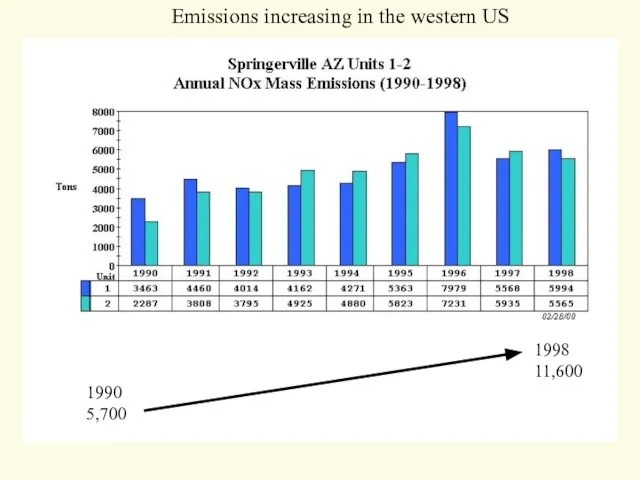
- 22. 1990 5,700 1998 11,600 Emissions increasing in the western US
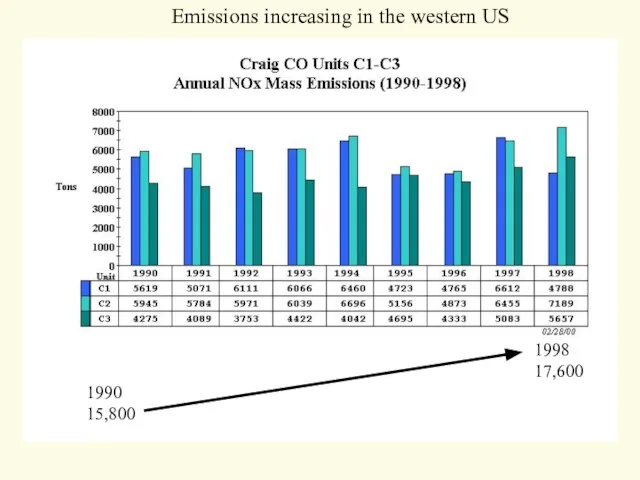
- 23. 1990 15,800 1998 17,600 Emissions increasing in the western US
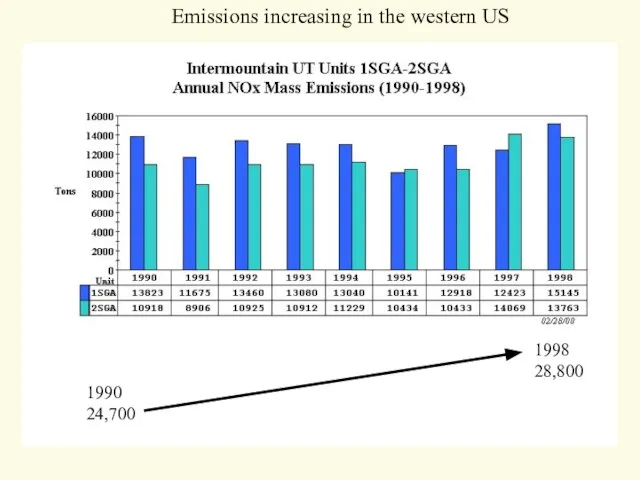
- 24. 1990 24,700 1998 28,800 Emissions increasing in the western US
- 25. Recent and current policies to reduce acid precipitation and Nitrogen emissions are shifting the problem from
- 26. Other types of air pollution The difference between stratospheric and tropospheric ozone Photochemical smog Inversion layers
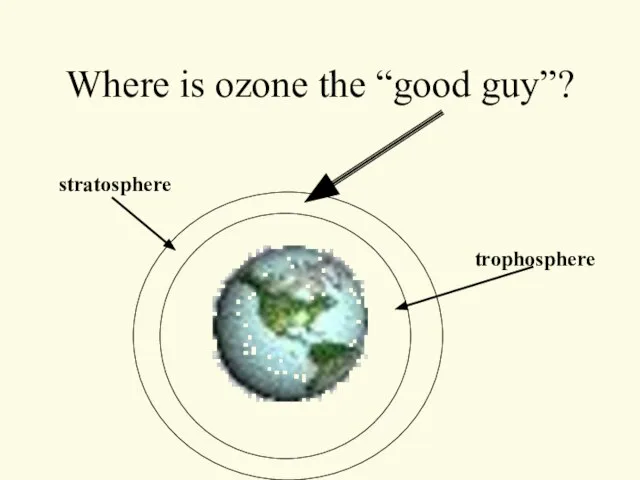
- 27. Where is ozone the “good guy”? trophosphere stratosphere
- 28. In the stratosphere…. Ozone blocks incoming Ultra-violet radiation Ultraviolet radiation Skin cancer Cataracts Plant Damage
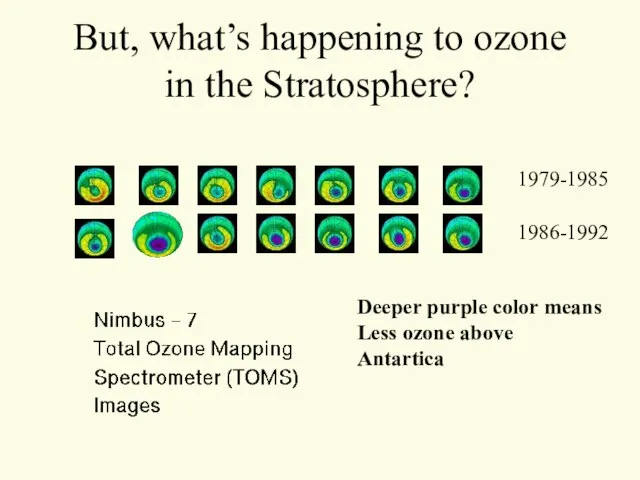
- 29. But, what’s happening to ozone in the Stratosphere? 1979-1985 1986-1992 Deeper purple color means Less ozone
- 30. Why? Chlorofluorocarbons (CFC) are very stable compounds that we produce at earth’s surface They migrate to
- 31. The Montreal Protocol has reduced use of CFC’s, but… Their long life span means that they
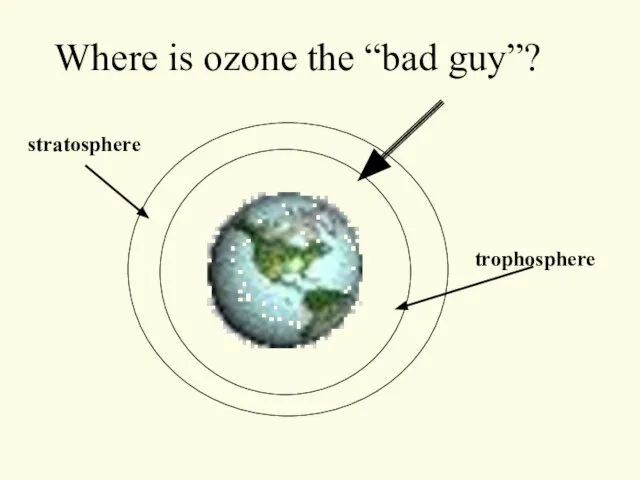
- 32. Where is ozone the “bad guy”? trophosphere stratosphere
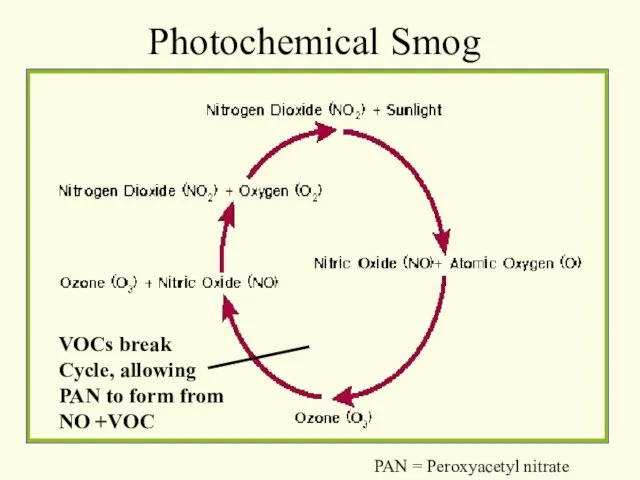
- 33. Photochemical Smog VOCs break Cycle, allowing PAN to form from NO +VOC PAN = Peroxyacetyl nitrate
- 34. Examples of Smog
- 35. NOx + VOC Ozone + Pan light reactants products Chemical Equation for Photochemical Smog PAN =
- 36. Where reactants come from NOx primarily from transportation VOC from a variety of sources, including refining,
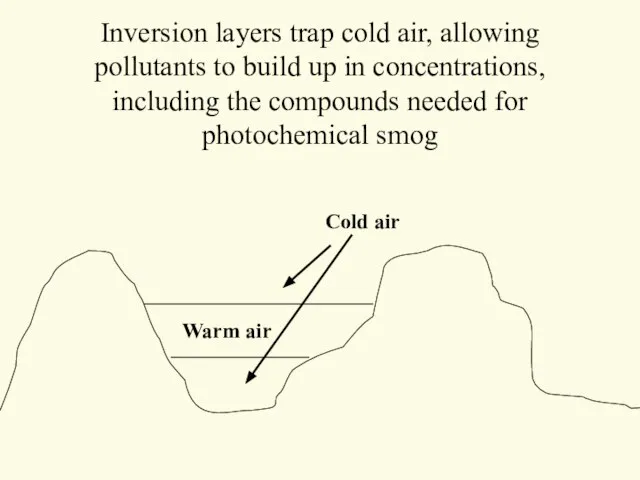
- 37. Inversion layers trap cold air, allowing pollutants to build up in concentrations, including the compounds needed
- 38. Ozone’s bad features Extremely reactive will burn leaves, lungs, synthetic compounds (e.g. rubbers, plastics) Because of
- 39. Humans depend on very small reservoirs of water for all our needs These reservoirs cycle/ turnover
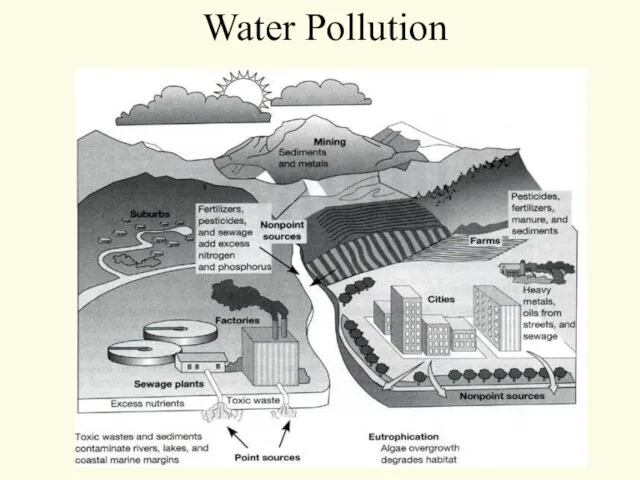
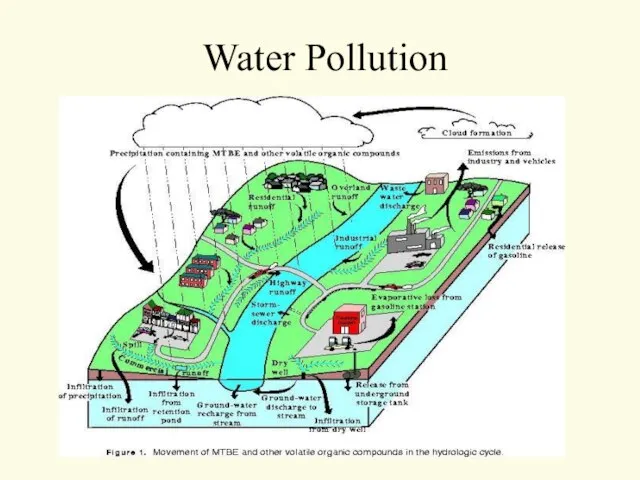
- 40. Water Pollution
- 41. Water Pollution
- 42. Water Pollution Two major classifications Point Source Non-point Source
- 43. Point Sources Single large source Can localize it to one spot Industrial Plants - Sewage pipes
- 44. Point Source - Example LUST - Leaky Underground Storage Tanks 22% of the 1.2 million UST
- 45. Point source examples
- 46. Non-point Sources Diffuse source or many smaller point sources Automobiles Fertilizer on fields
- 47. Non point source examples
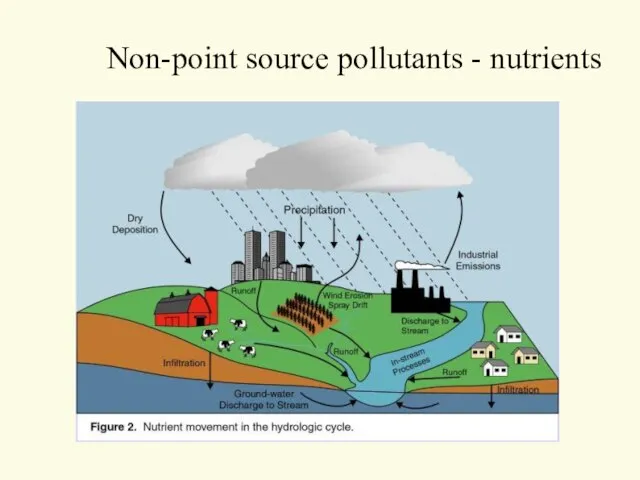
- 48. Non-point source pollutants - nutrients
- 49. End Lecture 4/22/03
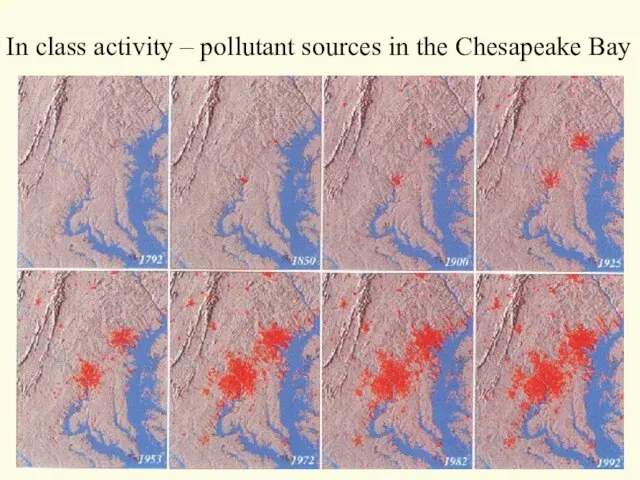
- 50. In class activity – pollutant sources in the Chesapeake Bay
- 51. The four main roles for class debate on 5/1/2003 Pacific Lumber Company /Maxxam Corporation The CEO’s
- 53. How does acid kill the fish? One way is mobilizing metals When all base cations are
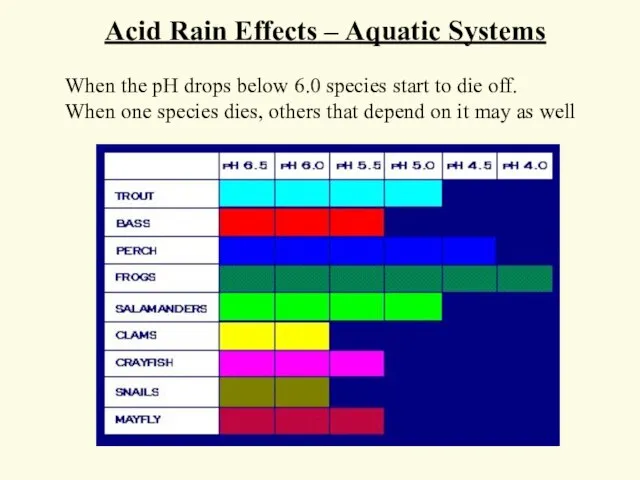
- 54. When the pH drops below 6.0 species start to die off. When one species dies, others
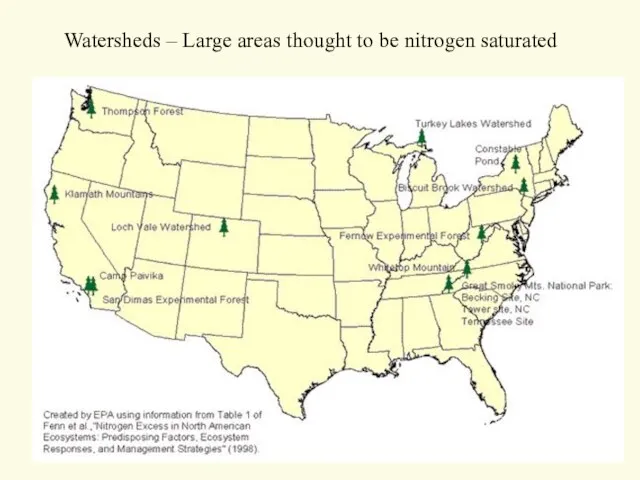
- 55. Watersheds – Large areas thought to be nitrogen saturated
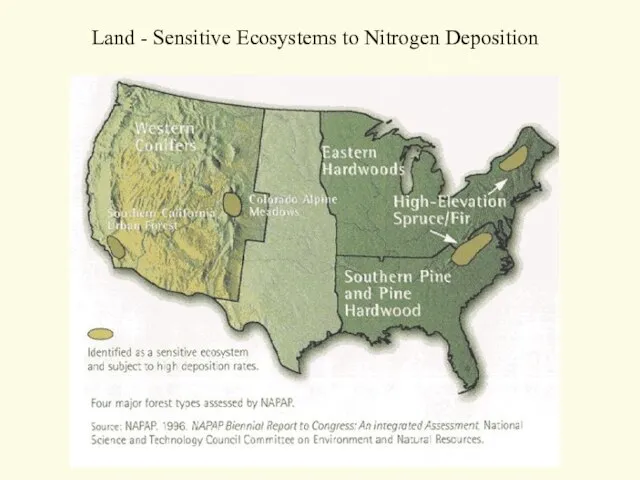
- 56. Land - Sensitive Ecosystems to Nitrogen Deposition
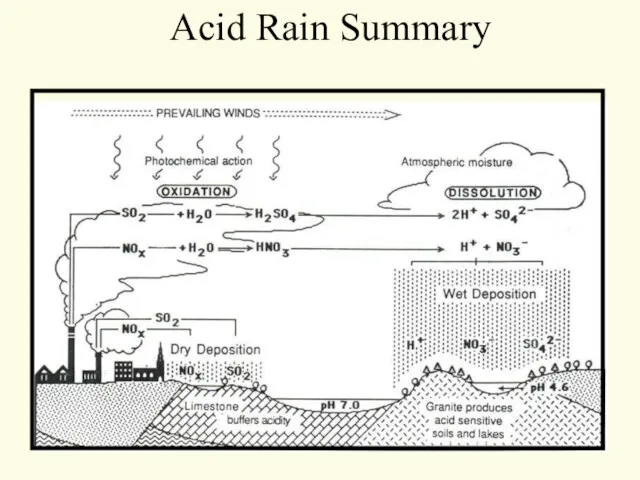
- 57. Acid Rain Summary
- 59. Скачать презентацию






























 Лексика на деловых встречах
Лексика на деловых встречах 效果图
效果图 Кейс Мошенничество в автостраховании и борьба с ним
Кейс Мошенничество в автостраховании и борьба с ним НЕВОДНЫЕ БУРОВЫЕ РАСТВОРЫ
НЕВОДНЫЕ БУРОВЫЕ РАСТВОРЫ «Певцы родной сторонки».
«Певцы родной сторонки». Презентация на тему Жук-носорог
Презентация на тему Жук-носорог Презентация на тему Многозначность понятия Наука
Презентация на тему Многозначность понятия Наука  ПОРТФОЛИО
ПОРТФОЛИО Англ презентация
Англ презентация Защита окружающей среды касается каждого
Защита окружающей среды касается каждого Ўзбекистонда ижтимоий диний бағрикенглик
Ўзбекистонда ижтимоий диний бағрикенглик Новые педагогические подходы в обучении студентов информатике в условиях модернизации образования
Новые педагогические подходы в обучении студентов информатике в условиях модернизации образования Тема урока: Создание декоративных изделий из древесины
Тема урока: Создание декоративных изделий из древесины Условия плавания тел.
Условия плавания тел. Бежит, гудит, В два глаза глядит, А станет – Ярко-красный глазок глянет.
Бежит, гудит, В два глаза глядит, А станет – Ярко-красный глазок глянет. Холодная война 9 класс
Холодная война 9 класс Классицизм в архитектуре
Классицизм в архитектуре Общая задолженность по оплате в ПИК ЕАСУЗ заказчиками Минздрава
Общая задолженность по оплате в ПИК ЕАСУЗ заказчиками Минздрава Произведения великих русских художников
Произведения великих русских художников Напиши мне письмо…
Напиши мне письмо… Вуглеводи
Вуглеводи Коррупция: зона особого внимания
Коррупция: зона особого внимания Как построить команду мечты
Как построить команду мечты Право, его роль в жизни общества и государства. Урок № 11
Право, его роль в жизни общества и государства. Урок № 11 Презентация на тему Сложение сил, направленных вдоль одной прямой (7 класс)
Презентация на тему Сложение сил, направленных вдоль одной прямой (7 класс) Окружающая среда и человек
Окружающая среда и человек Некоторые подсказки по разработке проектных предложений Чокушева Гульнара Национальный Офис программы Темпус в Кыргызстане Webs
Некоторые подсказки по разработке проектных предложений Чокушева Гульнара Национальный Офис программы Темпус в Кыргызстане Webs Всероссийский правовой диктант
Всероссийский правовой диктант