Содержание
- 2. Objectives of lecture –presentation: Study of different methods of nanosystems investigations
- 3. Plan of lecture –presentation: Lecture 5 1. Method of Electron Microscopy 2. Sonde Microscopy 3. Diffraction
- 4. The basic methods of nanoparticles’ sizes and some properties in gaseous phase determination: ionization by photons
- 5. Methods of study of particles on the surface: X-ray and scanning electron microscopy (information about sizes/forms
- 6. Methods of study of particles in volume: X-ray and scanning electron microscopy, electric conductivity, as well
- 7. 1. Method of Electron Microscopy Microscopy is the main method for determination nanoparticles’ sizes. They use
- 8. 1. Method of Electron Microscopy X-ray electron microscopy. Object in form of thin film is X-rayed
- 9. 1. Method of Electron Microscopy Scanning electron microscopy. This method is used mostly for study of
- 10. 1. Method of Electron Microscopy Several types of rays occur when electrons interact with the object:
- 11. 1. Method of Electron Microscopy The main value of this method – is that it is
- 12. 1. Method of Electron Microscopy Top: Scanning electron micrograph of a line-pair testing object with 50nm
- 13. 2. Sonde Microscopy In 1981 Binnig and Rorer created scanning tunnel microscope (STM) and in 1986
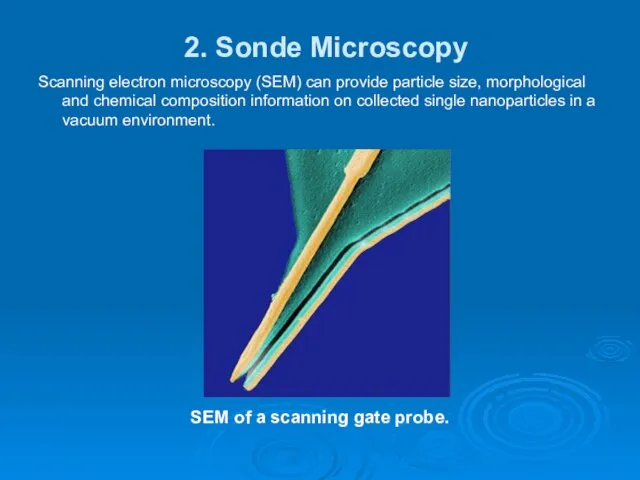
- 14. 2. Sonde Microscopy Scanning electron microscopy (SEM) can provide particle size, morphological and chemical composition information
- 15. 2. Sonde Microscopy General scheme of sonde microscope work Mechanic part of atomic-force microscope
- 16. 3. Diffraction Methods . These methods include diffraction of X-rays and neutrons and are less general
- 17. 3. Diffraction Methods . Neutrons diffraction. Neutron is the particle which due to its properties is
- 18. Plan of lecture –presentation: Lecture 6 1. Mass-Spectrometry 2. Photoelectron Spectroscopy
- 19. 1.Mass-Spectrometry Mass spectrometry (MS) is an analytical technique that produces spectra (singular spectrum) of the masses
- 20. 1.Mass-Spectrometry Mass spectrometry works by ionizing chemical compounds to generate charged molecules or molecule fragments and
- 21. Steps of mass-spectrometry: 1. Ionisation - Gaseous atoms of a particular element are bombarded with electrons
- 22. Schematics of a simple mass spectrometer with sector type mass analyzer. This one is for the
- 23. 2. Photoelectron Spectroscopy This method is based on the measurement of spectrum energies of electrons which
- 24. 2. Photoelectron Spectroscopy They determine energies of interactions of electrons and their energy levels in studied
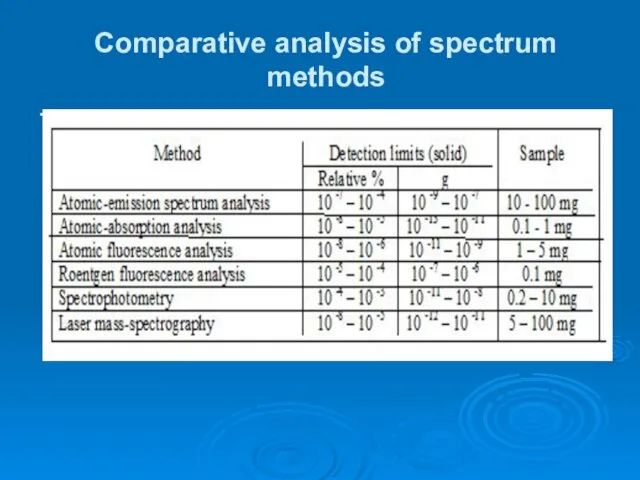
- 25. Comparative analysis of spectrum methods .
- 26. Check Yourself 1. What is the most significant for study of chemical interactions? 2. What are
- 27. Check Yourself 13. What restricts the resolution of conventional lenses? 14. Describe the method suggested by
- 28. Check Yourself 22. Which are the main requirements for crystals to be studied by roentgenography method?
- 30. Скачать презентацию

























 Федерализм
Федерализм Молоко
Молоко Производная и её геометрический смысл
Производная и её геометрический смысл Инвестиционно-финансовая группа
Инвестиционно-финансовая группа Беспроводная передача электричества
Беспроводная передача электричества Презентация на тему Кодирование графической информации
Презентация на тему Кодирование графической информации Использование деятельного подхода в ОД при формировании ОБЖ
Использование деятельного подхода в ОД при формировании ОБЖ Он-лайн система управленческого учета
Он-лайн система управленческого учета Типы реакций
Типы реакций Энергия солнца в Вашем Доме!
Энергия солнца в Вашем Доме! Московские лабораторииэкономики и технологии знанийпредставляют
Московские лабораторииэкономики и технологии знанийпредставляют Электроёмкость. Конденсаторы
Электроёмкость. Конденсаторы Измерение высоты здания разными способами
Измерение высоты здания разными способами Словообразования
Словообразования Восточная Европа
Восточная Европа Презентация на тему Инвестиции
Презентация на тему Инвестиции Получение наличных средств в сети круглосуточных банкоматов Сбербанка России и других банков по всему миру Безналичная оплата то
Получение наличных средств в сети круглосуточных банкоматов Сбербанка России и других банков по всему миру Безналичная оплата то Предпосылки петровских преобрахований
Предпосылки петровских преобрахований Люблю я Кавказ
Люблю я Кавказ Принцип построения сети ОКС №7. Режимы сигнализации
Принцип построения сети ОКС №7. Режимы сигнализации Программы обучения онлайн 2020
Программы обучения онлайн 2020 Дорогие ребята! Мы шагаем в проект «Среди миров»
Дорогие ребята! Мы шагаем в проект «Среди миров» Кашира
Кашира International Corporate Match Race 10-11 September 2011 Yacht Club of Greece
International Corporate Match Race 10-11 September 2011 Yacht Club of Greece Уголовный процесс Чешской республики
Уголовный процесс Чешской республики Парад студенчества 2018
Парад студенчества 2018 На все автомобили предоставляется Гарантия 2 года без ограничения пробега Прайслист действителен с 03.01.2012 г. Цены приведены в долла
На все автомобили предоставляется Гарантия 2 года без ограничения пробега Прайслист действителен с 03.01.2012 г. Цены приведены в долла О создании Восточно-Сибирского научно образовательного инновационного комплекса в Республике Бурятия
О создании Восточно-Сибирского научно образовательного инновационного комплекса в Республике Бурятия